Alzheimer’s disease is usually defined by the build-up of amyloid beta plaques and tau tangles. A new study, led by Prof Marc Aurel Busche and Dr Robert Ellingford (UK DRI at UCL) suggests that these hallmark proteins may inflict some of their most important damage by working together and disrupting the brain circuits that support memory. This helps to explain why targeting either toxic protein on its own does not bring therapeutic benefits, once both begin to accumulate. Published in Neuron, the study identifies a new mechanism that links amyloid beta and tau to this circuit failure by causing changes to synaptic connections within the brain.
The researchers found that Alzheimer’s does not simply reduce brain activity across the board. Instead, it disrupts patterns of activity within memory circuits linking the cortex and hippocampus in highly specific ways. These patterns matter because they allow brain cells to communicate effectively and support the processes needed for learning and memory. In superficial layers of the cortex, neurons lost a distinctive rapid firing pattern known as bursting, which is thought to be important for learning and memory. By contrast, deeper cortical layers and a region of the hippocampus known as the CA1, important for memory, showed broader reductions in neuronal firing. This suggests that different parts of the memory network fail in different ways in Alzheimer’s.
The study also helps explain how amyloid beta and tau may work together in Alzheimer’s. Amyloid beta pathology usually appears earlier in the disease, and the new findings suggest it may make brain circuits more vulnerable to the later arrival of tau. In the mouse models used in this study, the presence of toxic amyloid beta accelerated the emergence of tau-related dysfunction, supporting the idea that amyloid beta may lay the foundation for faster and more damaging effects of tau on brain activity. Similar amyloid beta related vulnerability was also seen in cerebral organoids – human neurons cultured in a dish to resemble the brain.
What surprised us was that Alzheimer’s pathology did not simply dampen activity across the brain. Different parts of the memory circuit were disrupted in different ways, and amyloid beta appeared to make those circuits more vulnerable to tau. That gave us a clearer picture of how the two hallmark proteins work together to undermine brain function.
Dr Robert EllingfordUK DRI Postdoctoral Researcher
At the centre of the findings is a problem at synapses, the points of contact and communication between neurons. The researchers found evidence that the combined presence of amyloid beta and tau is linked to reduced levels of GluN1 at the synapse, an essential component of NMDA receptors, which are important for learning and memory. This points to impaired synaptic NMDA receptor function as a key driver of the circuit disruption observed in the study. Importantly, the same synaptic change was also seen in post-mortem brain tissue of people affected by Alzheimer’s.
Perhaps most notably, once both toxic proteins began to accumulate in the mouse model, reducing only amyloid beta or tau was not enough to restore circuit firing to normal levels. Only by simultaneously reducing the levels of both toxic proteins were neuronal firing patterns rescued, which corresponded with improved performance on memory tests. The findings therefore provide a strong rationale for combination treatments that target both amyloid beta and tau, particularly once both are already present. They may also help explain why current amyloid beta lowering therapies appear to work best earlier in disease, when toxic tau is yet to become fully established.
Prof Marc Aurel Busche, senior author of the study, said:
“What is especially encouraging is that when we restored more normal activity patterns in these memory circuits, we also saw a blood marker of damage to neurons move back towards normal. That does not prove that circuit dysfunction directly causes later neurodegeneration, but it suggests that changes in brain activity may be an early and potentially reversible part of the disease process. In other words, brain function itself may be an important therapeutic target alongside amyloid beta and tau.”
Reference: Ellingford et al., Alzheimer’s disease pathology degrades an NMDA-receptor-dependent spontaneous-activity pattern in cortico-hippocampal circuits, Neuron (2026), https://doi.org/10.1016/j.neuron.2026.02.027
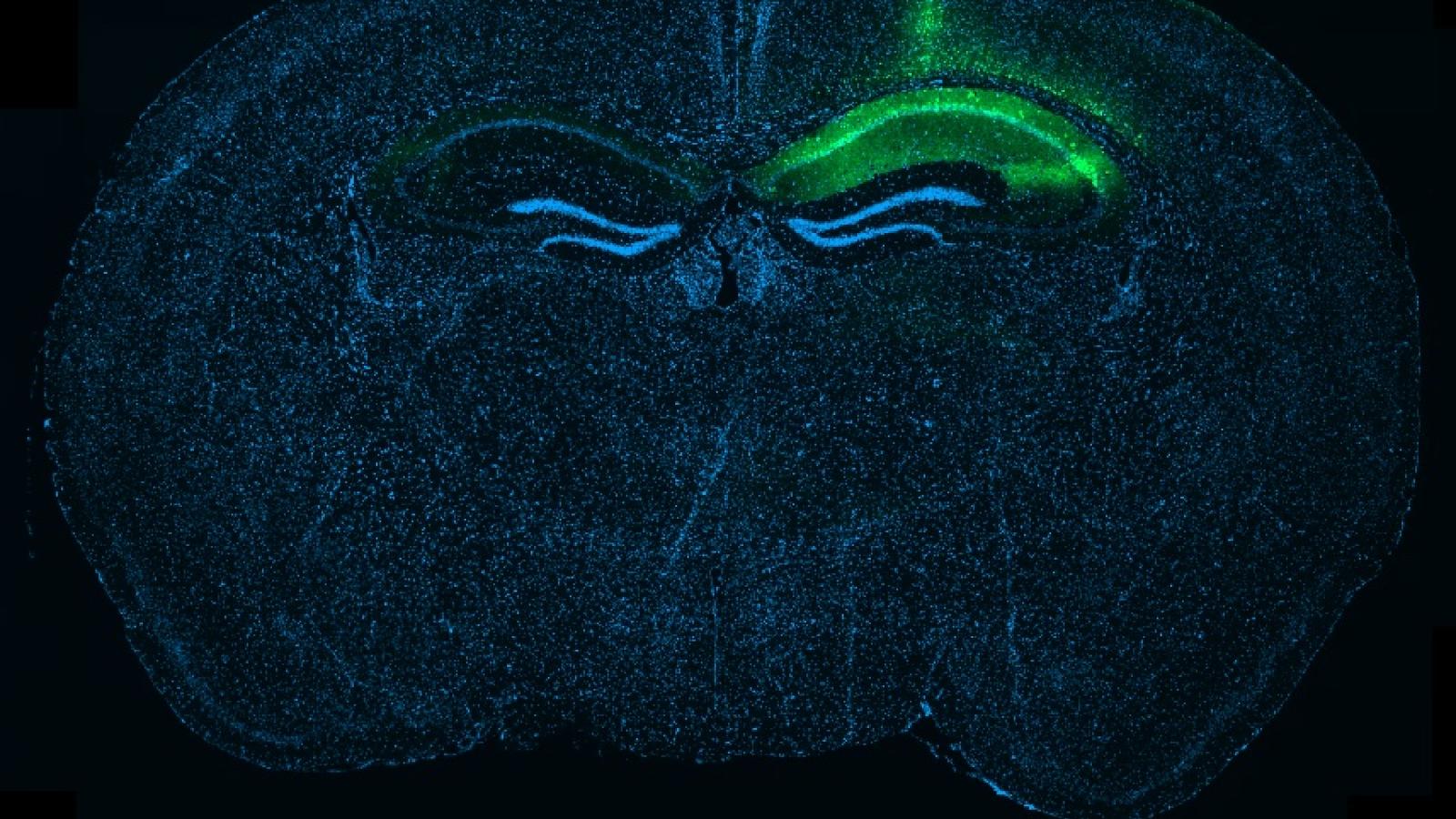
Banner image: Whole-brain section showing local tau expression in the hippocampus after a targeted injection, visible here as the green signal on one side of the brain. Credit: Robert Ellingford